Dr. Ahmad Shahzad
Founder | Lyallpur Diabetes Foundation
Consultant Diabetologist | Educator | Advocate for Preventive Care
Diabetes can affect many parts of the body, and one of the most serious yet often overlooked complications is Charcot foot. The condition is caused by the fact that nerve damage and poor circulation weakens the bones and joints of the foot, resulting in fractures, deformities and even disability, unless it is treated on time. Due to its blurred symptoms in the initial stages, like swelling, redness, and handiness, Charcot foot is often diagnosed at late stages when the problem is already in the progression stage. Learning about the Charcot foot, its formation, and preventive and curative steps is the key to maintaining long-term foot health in diabetic patients.
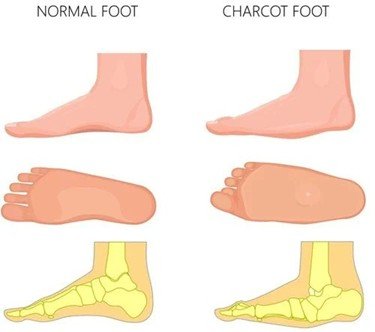
What is Charcot Foot?
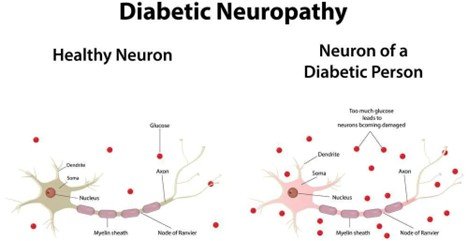
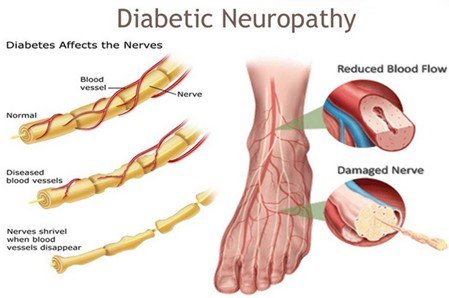
Charcot Foot is a progressive disorder whereby there is weakening of bones, joints, and soft tissues in the foot or ankle. It is mostly due to peripheral neuropathy (damage to the nerves) which is often found in individuals with diabetes, and this leads to loss of sensation in the foot. Because of this numbness, minor or repeated injuries go undiagnosed and unattended resulting in fractures, joint dislocations and subsequent foot deformity like the rocker-bottom foot, arch collapses. This malformation adds a risk of skin sores, ulcers, infection and even amputation unless treated aggressively.
The process occurs in phases starting with inflammation and swelling, bone fragmentation, and dislocation of the joints, bone healing, and deformities. Redness, warmth (usually with the affected foot being warmer than the opposite), swelling, and pain or soreness are common. Unattended, Charcot Foot may cause severe deformity and instability of the foot and ankle joints, making walking hard and predisposing to severe complications.
Causes and Risk Factors
Causes and Risk Factors include:
- Charcot foot is mostly seen in individuals with peripheral neuropathy (usually diabetes). The numbness in the foot causes injuries and recurring trauma to go undetected, initiating inflammation and the destruction of bones.
- History of prior diabetic foot issues like ulcers, surgery, amputation or trauma is the biggest risk factor. This preceding injury triggers the inflammatory event that causes Charcot arthropathy.
- Other significant risk factors include:
• Long duration of diabetes (over 10 years)
• Poor blood sugar control and complications like nephropathy and retinopathy
- Objective: Obesity that causes pressure and stress to the insensate foot.
• Age less than 60 years but with chronic diabetes.
• Previous foot surgeries and deformities
• Kidney disease and poor circulation
• Lifestyle factors like smoking, alcohol consumption, and sudden increases in activity in patients with neuropathy.
- Charcot foot may also be presented in other circumstances with nerve damage, including spinal cord injury, Parkinson, multiple sclerosis, or syphilis.
- Such a cascade can be initiated by minor, repetitive trauma or foot sprain in a person with neuropathy, resulting in bone erosion and deformity.
Symptoms and Warning Signs
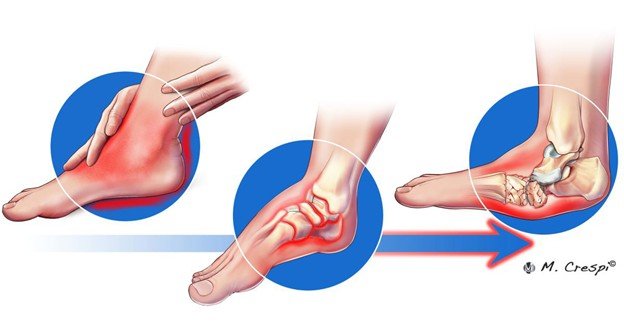
Symptoms and Warning Signs of Charcot Foot:
- It is the first indicators of Charcot foot and may at times be very sudden and sudden such as reddening, swelling of the foot or ankle and/or even at night. It can be much warmer around one foot than around the other.
- There can be mild pain or discomfort, and many patients have little or no pain despite severe injury because of nerve damage.
- The foot can also be swollen and reddish, and these symptoms may resemble infection or sprain, being often misdiagnosed.
- Later in the condition, the foot can become deformed with collapse of the midfoot arch (also known as rocker-bottom foot), curled toes as well as misalignment or dislocation of bones.
- The alterations of foot form may create irregular pressure sites, which result in blisters, wounds, or open ulcers. Such wounds are subject to infections and slow healing owing to reduced sensation and inadequate circulation.
- The foot swells and alters its shape, leaving patients with difficulty fitting into shoes.
- Charcot Foot Often, swelling and redness decrease with foot elevation and distinguishes it from infections where the redness and swelling do not disappear with elevation.
- Without treatment, deformity deteriorates, and this may lead to instability, walking problems and high chances of serious infections that may need amputation.
Stages of Charcot Foot Development
Charcot Foot develops through distinct stages, classically described in three main phases:
Stage I (Development or Fragmentation):
- The acute or the first stage is characterized by intense inflammation, redness, swelling and heat in the foot.
- The foot used to be hot in comparison with the other foot.
- Radiographs reveal destruction of bones with bone fragmentation, joint dislocation, fractures, and debris.
- This phase can take weeks to months, during which the foot is highly susceptible, and damage increases when weight bears are maintained.
Stage II (Coalescence):
- As the inflammation heals, swelling, redness, and warmth start to fade.
- The body begins to reabsorb bone debris and certain bone repairs take place.
- Radiographs reveal early bone sclerosis (hardness) and bone fusion beginning in selected locations.
- Foot stabilizing but deforming.
Stage III (Reconstruction or Consolidation):
- In this chronic stage, inflammation usually resolves, and the foot is no longer swollen or warm.
- Radiographs show bone healing with signs of remodeling, joint space narrowing, arthrosis (joint degeneration), and bone fusion.
- Despite healing, deformities—such as collapsed arches and foot misalignment—remain, often leading to a permanent change in foot shape.
- The foot becomes stable but structurally altered and prone to ulcers and other complications.
An additional Stage 0 has been described for clinical cases with signs of inflammation but no radiographic changes yet.
Diagnosis of Charcot Foot
Diagnosis of Charcot Foot involves clinical evaluation and imaging, with key components as follows:
Clinical Examination:
- Typical signs include edema (swelling), redness (erythema), and high temperature of the involved foot or ankle.
- Foot is usually warm, relative to other foot, occasionally by as much as 2C.
- The disorder is often free or with minimal pain because of peripheral neuropathy, yet patients complain of mild discomfort.
- A useful bedside exam is to raise up the affected foot when the patient is in the lying position with his feet raised up 5-10 minutes; swelling and redness in Charcot Foot will generally reduce, but swelling related to infection will likely not.
- Peripheral neuropathy should be evaluated with the help of the Semmes-Weinstein 10-g monofilament to determine absence of protective sensation.
- Infection should be assessed; foul odor, purulence, or gangrene are indicators of infection, not pure Charcot.
Imaging:
- X-rays (radiographs): the first and most common imaging modality to consider fracture, bone destruction, joint dislocation, and changes in alignment.
- Early acute Charcot can be missed in X-rays and not detected until weeks later; repeat X-rays in 2 weeks can be useful.
- MRI can help identify bone marrow edema and subtle changes early and distinguish between Charcot Foot and osteomyelitis, particularly when ulcers or infections are suspected.
- Bone scans and leukocyte scintigraphy may occasionally help differentiate Charcot and infection, but are limited.
Laboratory Tests:
- Blood glucose testing and HbA1c to assess diabetes control.
- Inflammatory markers like ESR and CRP can help differentiate infection (usually elevated) from Charcot (may be normal or mildly elevated).
Treatment and Management
Treatment and Management of Charcot Foot:
- Treatment involves mainly immobilization and offloading of affected foot to prevent further damage, inflammation and enable healing. This is normally done with a non-removable below-knee Total Contact Cast (TCC) or an irremovable walker cast. Immobilization can last several months before the acute phase heals.
- Patients should also not bear weight on the affected foot much, which may necessitate the use of mobility aids like crutches or wheelchairs.
- Follow-up and re-cast (every week or biweek) might be required regularly to make swelling corrections and follow up on healing progress.
- Patient education on the condition, the significance of strict compliance with offloading and immobilization, and the control of blood sugar is essential to successful work.
- After the acute phase is over, patients switch to individual orthotics, special shoes, or braces to support foot defects and avoid ulcers or disease relapse.
- Surgical intervention can be discussed in severe deformation cases, instability, or when ulcers and infections occur. Surgery may consist of realignment and stabilization of foot bones or in severe cases, amputation.
- Multidisciplinary care, such as endocrinologists, podiatrists, orthopedic surgeons, and physical therapists, is frequently included as well.
- Psychological and emotional support can be helpful because anxiety and depression are frequent in patients.
- It should maximize management of diabetes, peripheral neuropathy, and other risk factors to avoid progression or relapse.
- The routine use of drugs such as bisphosphonates in Charcot foot is not supported by strong evidence.
- Prevention is also useful, like the use of the correct footwear on the opposite foot because both feet can be involved in Charcot.
Bottom Line
Charcot Foot: A Serious Complication of Diabetes is an illness that needs to be identified and managed at an early stage to avoid long-term disability. It may cause extreme deformities, infections, and even amputation but with early diagnosis, off-loading, and continued foot care, most of them are preventable. Foot reviews, suitable footwear, and routine medical engagements are fundamental components of mobility and the general quality of life among individuals with diabetes. Through awareness and active management, we can minimize the risks and assist people to live healthy and active lives in spite of the complications of diabetes.