Dr. Ahmad Shahzad
Founder | Lyallpur Diabetes Foundation
Consultant Diabetologist | Educator | Advocate for Preventive Care
As we know, diabetes can have an impact on blood sugar and general metabolic wellbeing, but that is just the tip of the iceberg. Bone health is a field that is not given much attention. As per the research, individuals with diabetes have a greater risk of fractures, low bone density, and low bone strength. It is important to understand the impact of diabetes on the skeletal system to prevent this condition, detect it early and improve the long-term health outcomes.
Understanding the Link Between Diabetes and Bone Health
Diabetes causes serious complications in bone health, bone density, and bone quality, predisposing individuals to fracture. Type 1 diabetes (T1DM) and type 2 diabetes (T2DM) both cause bone problems, albeit in slightly different ways.
Impact on Bone Density and Structure
· T1DM is typically linked to reduced bone mineral density (BMD), which disables bone formation. This is attributed to hyperglycemia leading to accumulation of advanced glycation end products (AGEs) which destroy bone cells, disrupt the activity of osteoblasts (bone-building cells) and augment bone resorption by osteoclasts. T1DM also depletes growth factors like insulin-like growth factor-1 (IGF-1), which also inhibits bone growth and bone remodeling.
· T2DM, ironically, is commonly characterized by normal or even better BMD but worse bone quality. The prolonged hyperglycemia and related metabolic alterations worsen the organic structure and biomechanical characteristics of the bone, making bones weaker despite increased density. The bone-forming cells are also adversely affected by increased bone marrow fat and inflammation in T2DM.
Increased Fracture Risk and Healing Complications
· T1DM and T2DM patients are more likely than non-diabetics to suffer a fracture. This is not necessarily attributed to structural integrity of the bone but also due to complications of diabetes like neuropathy and muscle weakness, eye issues and frequent falls which increase the risk of fracture.
· The recovery of fractured bones in diabetics may be delayed due to impaired bone cell activity, reduced progenitor cells and poor vascularization of fracture locations. Long-term hyperglycemia may have permanent adverse effects on bone cell activity, which impairs proper repair.
Clinical and Treatment Considerations
- Prevent or reduce diabetic bone disease through good glycemic control. Certain antidiabetic drugs (e.g., metformin) are potentially better since they have fewer adverse effects on bone health, whereas other drugs (such as thiazolidinediones) can increase bone loss.
- Therapies focused on the enhancement of bone density and quality, including anti-resorptive medications (e.g., alendronate), bone-forming (e.g., intermittent PTH in T1DM), and vitamin K2 and vitamin D3 supplementation in T2DM, have potential in the treatment of bone complications in diabetes.
- As well as acting to regulate blood sugar, insulin therapy has direct positive effects on bone formation by stimulating osteoblastic activity.
Mechanisms: How Diabetes Affects Bones
Diabetes affect bone health through several interconnected mechanisms centered around hyperglycemia, insulin deficiency or resistance, and inflammation, which alter bone cell function, bone matrix quality, and bone remodeling processes.
Key Mechanisms
- Hyperglycemia and Advanced Glycation End Products (AGEs): Chronic high blood sugar causes the development of AGEs that build up in bone collagen. This deposition makes the collagen matrix weaker and less durable, which reduces the mechanical strength and quality of bone. AGEs also induce bone progenitor cell apoptosis (death), osteoblast differentiation and function, and osteoclastic activity, resulting in elevated bone resorption.
- Impaired Osteoblast Function and Reduced Bone Formation: In type 1 diabetes (T1DM), there is a lack of insulin and growth factors such as insulin-like growth factor 1 (IGF-1) that inhibits the differentiation of mesenchymal stem cells (MSC) to osteoblasts. New bones are formed by osteoblasts, and thus their dysfunction reduces bone formation and bone mineral density (BMD). Repeated hyperglycemia suppresses important genes and proteins in osteoblast maturation and activity as well.
- Disturbed Bone Remodeling Dynamics: Diabetes causes disequilibrium in bone remodeling. The AGEs and the inflammatory cytokines enhance the activity of osteoclasts (bone resorption cells) and reduce the activity of osteoblasts, impairing normal bone turnover. This leads to poorer bone structure and fragility even with normal or even higher BMD particularly in diabetes type 2 (T2DM).
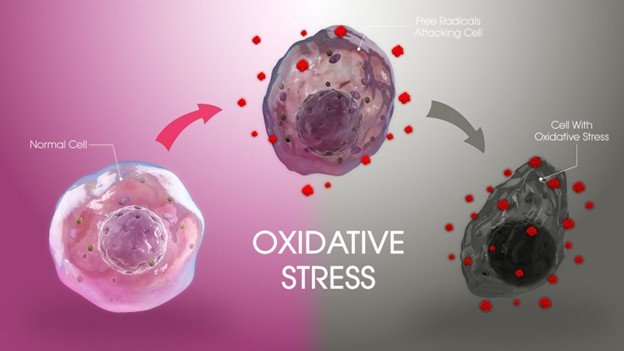
- Inflammation and Oxidative Stress: High blood sugar increases oxidative stress and proinflammatory pathways (e.g., NF-kB) and releases cytokines, such as TNF-a and IL-6. Such cytokines also suppress osteogenic differentiation of bone marrow stem cells and bone repair. Ironic inflammation also leads to bone quality and healing capacity.
- Bone Marrow Adiposity and Cellular Changes: In T2DM, bone marrow fat accretion leads to release of free fatty acids and pro-inflammatory cytokines, which worsen insulin resistance and suppress osteoblast activity. Bone marrow population of MSC also reduces which hinders the formation of new bone cells and restoration of broken bones.
- Vascular and Healing Losses Diabetes-induced loss of MSC viability and function also impairs vascularization of fracture sites, resulting in delayed bone healing, increased incidence of non-union fracture, and postoperative complications in diabetic patients.
Simply put, diabetes impairs bone health by exerting hyperglycemia-induced biochemical effects, inflammation, and altered stem cell processes that increase the risk of fracture and decrease bone repair potential.
Increased Risk of Fractures in People with Diabetes
Individuals with diabetes, especially type 2 diabetes (T2DM), are at a higher risk of developing bone fractures because of multifactorial etiological factors other than bone mineral density (BMD). This increased risk of fracture is due to several factors:
Risk Factors
- Bone Quality Impairment: Although many T2DM patients have normal or increased BMD, their bone quality is impaired by the deposition of advanced glycation end products (AGEs), low bone turnover, and alteration of bone microstructure, which impair bone tissue mechanical strength and strength.
- Duration and Severity of Diabetes: Long diabetes time (particularly 15 years and above) and inadequate glycemic control raise the risk of fractures. Fracture risk is also linked to insulin therapy which often is a sign of severe or advanced disease.
- Age and Body Mass Index (BMI): Elderly diabetic patients are at risk of getting a fracture. There is a significant increase in the risk of fractures in diabetic patients with a low BMI (<25 kg/m2), and a moderate effect of higher BMI.
- Diabetes Complications and Hypoglycemia: Diabetes-related conditions, including peripheral neuropathy, muscle weakness, impaired vision, and hypoglycemia (particularly because of drug use) are known to put patients at risk of falls and fractures.
- Medications: Thiazolidinediones, SGLT2 inhibitors, and insulin are some diabetes therapies that have been associated with an increased risk of fractures either via their impact on bone metabolism or by raising the risk of falls.
- T2DM patients experience higher fractures at the hip, vertebrae, and feet, but not always wrists, shoulders, or ankles.
Additional Factors Contributing to Poor Bone Health in Diabetes
Additional factors contributing to poor bone health in people with diabetes go beyond changes in bone mineral density (BMD) and include both skeletal and extra-skeletal influences:
- Advanced Glycation End Products (AGEs) Accumulation Hyperglycemia increases AGEs formation, changes bone collagen, making bones more rigid, fracture-prone, and brittle. This affects bone quality without BMD.
- Raised Fall Risk: Diabetes complications like peripheral neuropathy, impaired balance, muscle weakness, vision issues, and hypoglycemic episodes predispose one to fall, significantly contributing to the risk of fractures.
- Negative Effect of Some Medicines: Some antidiabetic medications (e.g. thiazolidinedions) have been shown to increase the risk of fractures by either impairing the formation of bone or enhancing its resorption. There are certain reservations about SGLT2 inhibitors that are mixed. Insulin therapy is anabolic to bone on its own, and usually a sign of advanced diabetes that correlates with bone fragility.
- Physical Inactivity: Diabetes complications reduce mobility, which slows down mechanical loading of bones and increases bone loss and skeletal weakness.
- Inflammation and Oxidative Stress: Chronic inflammation caused by diabetes and oxidative stress destroys bone cells, alters the work of osteoblasts and stimulates bone resorption.
- Finally, The Bone Marrow Composition Changes Type 2 diabetes alters the balance of bone marrow fat content which disrupts normal bone remodeling by preventing osteoblast differentiation and enhancing adipogenesis in bone marrow.
- Vitamin D Deficiency and Calcium Imbalance: Diabetes may be associated with a lack of vitamin D and poor calcium metabolism to weaken bone mineralization and strength.
- Comorbid Conditions: Diabetes may be comorbid with many conditions, including kidney disease, autoimmune diseases (e.g., celiac disease, Graves’ disease), hormone imbalance, and eating disorders, all of which may lead to poor bone health.
- Genetic Factors: Genetic polymorphisms in collagen synthesis genes and vitamin D receptor genes can also be involved in bone fragility in diabetes.
You may also like to read: Connection between Diabetes and Cognitive Decline
Strategies for Protecting Bone Health in Diabetes
Treatment of bone in diabetic individuals is a multifaceted process that involves proper glycemic control, lifestyle changes, nutritional interventions, drug-specific selection and, in some cases, bone-specific therapy.
Key Strategies
- Optimal Glycemic Control: Good blood sugar control minimizes the development of damaging advanced glycation end products (AGEs) in bone tissue and the risk of diabetic complications such as neuropathy and hypoglycemia that elevates the risk of falls and fractures. It is important to prevent hypoglycemic episodes by avoiding frequent ones.
- Lifestyle and Physical Activity: Resistance and weight bearing exercises (walking, strength training) promote bone density and bone strength. Exercise also enhances muscle tone and stability, which prevent falls. Quitting smoking and alcohol reduction are essential because these two habits undermine bones and predispose them to fractures.
- Nutritional Support: Optimal consumption of calcium and vitamin D is the key to good bone health. When the dietary intake is inadequate or vitamin D is low, supplements may be required especially in type 1 diabetes and the elderly with type 2 diabetes. Proteins are useful to maintain bones and muscles.
- Drug prescribing and Reimbursement: Select bone-neutral or bone-positive diabetes agents, including metformin, GLP-1 receptor agonists, and DPP-4 inhibitors. Thiazolidinediones (TZDs) should be avoided and SGLT2 inhibitors should be monitored because they may increase the risk of fractures. Insulin is to be administered with caution to prevent hypoglycemia.
- Bone-Targeted Therapies: In patients with osteoporosis or with a high risk of fracture, antiresorptive medications such as bisphosphonates (e.g., alendronate) and anabolic therapies such as teriparatide (PTH 134) have demonstrated effectiveness in enhancing bone density and fracture risk. Vitamin K2/D3 combination therapy has been demonstrated to induce bone formation in diabetic bones.
- Bone Health Check: It is essential to monitor bone mineral density (BMD) and bone turnover, as well as assess fracture risk, to ensure prompt treatment. Improved risk prediction of fractures in diabetes might be achieved by newer imaging methods and customized risk models.
- Make improvements to Fall Risk and Comorbidities: Fall management: treat diabetic neuropathy, visual issues, and muscle weaknesses to decrease falls. Vitamin D deficiency and other comorbidities of bone health are significant aspects of care that require screening and treatment.
Final Thoughts
Finally, diabetes does not only influence the level of blood sugar but also has an important effect on the strength of bones and general skeletal health. Diabetes demonstrates the need to make proactive care a priority by raising the risk of low bone density, fractures, and slow healing. It can be avoided by keeping blood sugar levels, consuming a balanced diet with bone strengthening nutrients, exercise, and regular examinations. People with diabetes can minimize the complications and help secure a healthier and stronger future with the right preventive measures.