Sundus Bukhari1, Mutayyaba Majeed2, Maria Liaqat3, Sana Shakeel1, Maria Muddassir4*,Faheem Hadi5, Tahir Maqbool11Institute of Molecular Biology and Biotechnology, The University of Lahore. Lahore, 2Department of Physiology, Independent Medical College. Faisalabad,Department of Pharmacology, University College of Medicine and Dentistry, 3The University of Lahore. Lahore,4M. Islam Medical & Dental College, Gujranwala, 5Faculty of Medicine and Allied Health Sciences, The Islamia University of Bahawalpur. Bahawalpur.
Abstract
Background: Worldwide, Hepatitis C virus (HCV) infection is a major public health concern. Twelve weeks following treatment completion, the persistent virologic response is known as SVR. The DAA (Direct-acting antiviral therapy) remains a challenge.Objective: To determine the demographic and biochemical parameters responsible for the failure of antiviral therapy (sofosbuvir and daclatasvir) associated with hepatitis C sustained viral response.Methods:Thiscross-sectional studyis based on Electronic Medical records online through a portal provided by the Government of Punjab Pakistan between December 2023 and July 2024. Data of 50 Hepatitis C screened positive patients were registered after theapproval from The University of Lahore’s ethical review board. The Patients underwent biochemical Investigations and HCV RNAPCR. Patients were given three months of combined therapy of Daclatasvir 60 mg and Sofosbuvir 400 mg. Monthly follow-ups were taken and Sustained virology response (SVR12) was determined, whereby SVR is the sustained virologic response 12 weeks after completing treatment.Results:Significant association has been found between DAA treatment and creatinine level, hemoglobin level and patient status. Demographically, mean age was 16.6±0.5 years, 19/50 (38.0%) were male and 31/50 (62.0%) were females. Concerning respondents, 66.0% were cured and 34.0% had a relapse. SVR was achieved in 66% cases. HCV patients responded well to a combined antiviral therapy.Conclusion:A strong correlation existed between DAA treatment, patient status, hemoglobin level, and creatinine level. Combined antiviral therapy showed good results for achieving SVR rates.Key words:Hepatitis C virus, DAA therapy, infection, daclatasvir, sofosbuvir.
Introduction
Despite widespread advocacy regarding the prevention of Hepatitis C and public health programs, the incidence of Hepatitis C is rising. HCV is a single-stranded RNA virus in the Flaviviridae family.1In adverse cases, HCV can cause permanent damage to the liver, including liver cirrhosis or hepatocellular carcinoma and sometimes even death.2 The primary transmission route for HCV dissemination is exposure to contaminated blood products or blood, unsafe injection practices and occupational exposure.3Hepatitis C infection occurs all around the globe.4,5According to the WHO report, almost 3% of the total population of the world has been infected with hepatitis C and more than 180 million people are chronic carriers of HCV virus and at increased risk of developing hepatocellular carcinoma and liver cirrhosis.4,5Recently, the DAA use for HCV treatment has led to a significant improvement in the rates of SVR in patients with genotype 1 HCV infection.6However, it can lead to a resistant virus selection when DAA is used alone. The NS5A protein, the NS5B RNA-dependent RNA. polymerase, and the NS3/4A protease are the three primary viral targets that are now the focus of HCV replication inhibition.7This rational combination have overcome the drugresistance challenge and improved the efficacy and safety profile with fewer adverse effect and drug-drug interaction.8The government of Pakistan has started national and provincial hepatitis prevention and control initiatives, which include screening for and treating HCV-infected individuals. However, the frequency of HCV is higher in spite of all these measures.9However, data are lacking on the effectiveness of these interventions; therefore, our aim is to investigate the possible demographic and biochemical parameters responsible for the failure of antiviral therapy associated with hepatitis C sustained viral response.9In Pakistan, the first-line treatment has been changed to the new DAA comprises Daclatasvir and sofosbuvir with or without ribavirin.This has improved patient adherence and have a better safety profile.10Daclatasvir is an HCV NS5A replication complex inhibitor that is used in combination with sofosbuvir, with or without ribavirin. The 12-week regimen of sofosbuvir and daclatasvir in apatient with genotype 1 and 3 HCV infection has shown a high SVR, irrespective of prior treatment experience.11The primary objective of this study is to identify the biochemical and demographic factors that contribute to the failure of antiviral treatment in relation to the sustained viral response in hepatitis C.
Methods
It was a cross-sectional study and Non-probability sampling technique was used to collect samples. The data of 50 Hepatitis C screened positive patients were registered on ElectronicMedical records online through a portal provided by the Government of Punjab Pakistan between December 2023 and July 2024. Informed consent was taken from patients or patient attendants. Demographic data was taken on the questionnaire. The study includedpatients 18 to 60 years of age who were Seropositive for HCV antibodies, HCV genotype 1 or 2 or 3 infections, patients with prior treatment and who will be confirmed failure during or after treatment with Daclatasvir and Sofosbuvir, patients havingAST toPlatelet ratio index (APRI)of ≤2, and who were diagnosed Cases of Chronic liver disease. Patients having chronic liver disease other than HCV infection, critically ill patients and those having co-infection with HIV or Hepatitis B virus were excluded from this study.A total of 80 HCV patients were referred patients from Hospital OPD and were screened under supervision on Rapid Diagnostic Kits (RDT). Hepatitis C screened positive patients were registered on Electronic Medical records online through a Portal provided by the Government of Punjab Pakistan. The Patients underwent biochemical Investigations like Hemoglobin, Liver function tests, Renal function tests, PT, and APTT. The patient’s sample for HCV RNA PCR was taken and sent to Government Punjab Headoffice Lab. Following the collection of PCR reports, the patients were given three (3) months of DAA (Direct Antiviral Agents) which will include Daclatasvir 60 mg and Sofosbuvir 400 mg.Monthly follow-ups was taken which include General body response andbiochemical tests like CBC and LFT. PCR will be taken after three months of End Treatment response (SVR12).The data was analyzed using Statistical package for social sciences (SPSS) version 23.0. Quantitative variables were expressed as mean ± standard error of the mean (S.E.M). The statistically significant value accepted with p<0.05. Relative results of all three groups were obtained and subject to t-test, and anova as per requirement.
Results
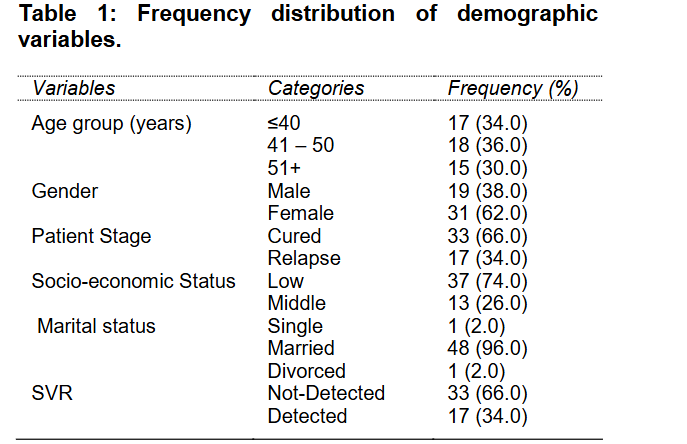
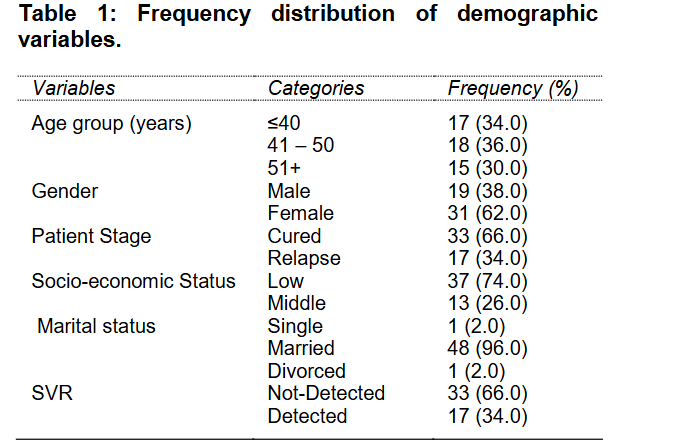
In this study, a data of 50 participants have been analysed. Frequency distribution of respondents regarding different demographic characteristics is given in Table-1. Mean age was 16.6±0.5 years. 17 (34.0%) respondents were of age less than or equal to 40 years, 18 (36.0%) were from 41 years to 50 years and 15 (30.0%) were of age greater than 50 years. Out of 50 respondents, 19 (38.0%) were male and 31 (62.0%) were females. Among respondents, 33 (66.0%) were cured and 17 (34.0%) were relapse. The SVR test showed that out of 50 patients, SVR was achieved in 66% cases.
The association between SVR and demographic variables has been determined by using the Pearson Chi square test. No significant association has been found between age of the participant andSVR (p=0.198). This means that there is no effect of age in SVR detection. No significant relationship was found between SVR and gender of the patient (p=0.806). This shows that SVR (detected or not detected) and gender of the respondents were independent. Significant association has been found between SVR and patient status (<0.001). Overall, no significant relationships have been found between age, socioeconomic status, gender, marital status and SVR(Table-2).
Two sample T test was applied to check the difference between patients where SVR was detected or not detected. In 33 patients SVR was notdetected and in 17 patients SVR was detected. With respect to urea in the patients, it can be concluded that the mean difference between SVR not-detected and SVR detected patients is non-significant. Hb value for patients where SVR was not-detected found higher. As p-value >0.05, therefore, we concluded that the PCR value in both groups was statistically non-significant(Table-3). According to the study’s findings, HCV patients responded effectively to combination therapy consisting of sofosbuvir and daclatasvir. Sixty-six percent of the participants in this trial had eradication of HCV successfully.
Discussion
Chronic hepatitis C infection is a major public health issue in Pakistan. It is one of the leading causes of mortality and morbidity associated with liver disease. It is believed that HCV-infected patients are more prone to failure of treatment. While the issue related to the efficacy of DAA treatment in HCV-infected patients has been explored extensively. Identifying the predictors of HCV treatment outcomes before the DAA treatment initiation is important in finding high-risk patients and alerting healthcare personnel to form a strategy to address treatment barriers. Identifying predictors of treatment outcomes help in reducing healthcare cost by preventing unnecessary retreatment and public health outcomes related to unattained SVR.A few studies only found the factors that affect the treatment outcome. In this study, 50 patients with genotype 1, 2 and 3 were enrolled and were treated with daclatasvir and sofosbuvir. The treatment duration was 12 weeks in treatment-experienced patients. The findings of this study reveal that sofosbuvir and Daclatasvir combination therapy worked well for HCV patients. In this study, 66% of patients have achieved successful HCV eradication. These findings are in accordance with the study carried out by Fontaine et al, in which a high SVR rate have been achieved in a patient given combination therapy of sofosbuvir and daclatasvir. In another study carried out in Egypt, more than 18000 patients suffering from HCV infection have achieved the SVR-12 rate of 95%.12An important finding of this study is that host-related factors that affect treatment outcomes, such as gender, age, marital status, and socioeconomic status weresignificant in this study cohort. Regarding age, only a few previous studies have shown the association between age and SVR rates in DAA regimens. The similar findings have been found in a study in which albumin level, age and gender are independent factors with DAA treatment.13In some studies, very little association between age and SVR rates has been observed.14This study failed to determine any significant difference between SVR and AST, ALT, albumin level, urea, prothrombin time and APRI score. This is inconsistent with the findings of yet another study whereby a significant difference has been found between platelet counts and SVR.15This comes in the same line with previous studies but different from the findings in which no significant difference has been found between platelet count and SVR.16The study shows that the sofosbuvir and daclatasvir combined therapy provided high rates of cure. This finding is consistent with the study conducted in which the DAA regimen proves to be effective with high cure rates and adverse effect with low incidence.17This finding is also in line with the study carried out by Pol et al. in which it is demonstrated that sofosbuvir and daclatasvir combination therapy had high antiviral potency.
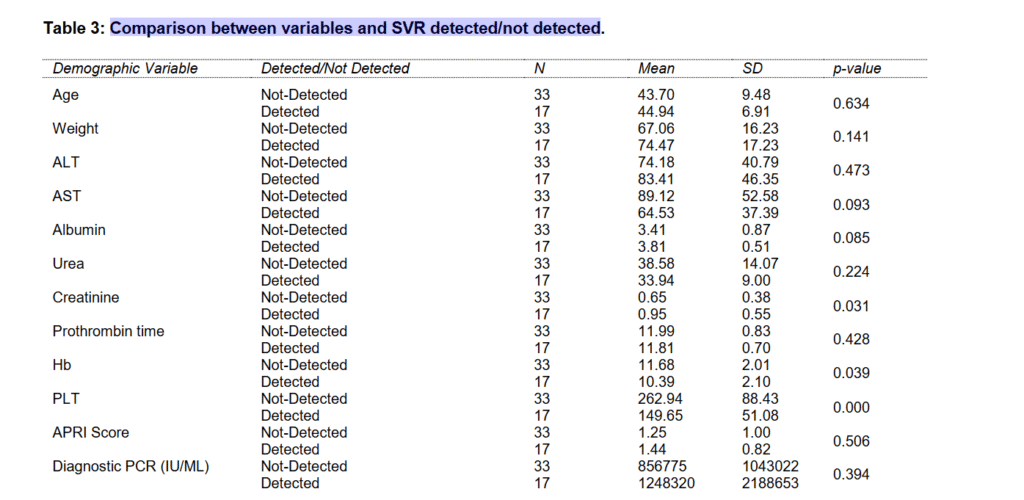
The finding shows that a significant difference is found betweencreatinine and SVR. Previous studies also show that creatinine levels are significantly lower in individuals who show treatment failure. This finding is consistent with the previous studies’ findings that haemoglobin is an important predictor of DAA regimen.22For example, in one retrospective study carried out on 152 patients with HCV infection, almost 15% experienced anemia.19The findings have shown that Hb level before the treatment of DAA has no impact on virologic response but is associated with increased serum creatinine.23The finding of this study shows no significant difference between albumin level and SVR rate. This finding is not in line with the previous studies in which it was found that higher albumin is related to the achievement of SVR.20Another important finding of this study is that Sofosbuvir and daclatasvir combination therapy has found to be effective in patients with genotype 3 HCV infection.21There were few limitations to this research study. First, this study uses the secondary data from the hospital department, and thus, not all variables were available for analysis. Another limitation of this study is the small sample size. More comprehensive data should be obtained for in depth mechanism and making clear interpretation regarding the current association.
Conclusion
Age, gender, marital status and socioeconomic status have been found to be independent factors in achieving SVR rate. The combined daclatasvir and sofosbuvir regimen are recommended in treating genotype 1, 2 and 3HCV-infected patients. Based on the findings of this study, combined sofosbuvir and daclatasvir treatment found to have favorable outcomes of achieving SVR rates in patients with chronic HCV infection.
Funding: None.
Availability of Data: The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethical Approval: The ethical review board of University of Lahore’s approved the study via letter no.IMBB/BBBC/22/11.
Conflict of Interest: None declared
References
1.Tabata K, Neufeldt CJ, Bartenschlager R. Hepatitis C virus replication. Cold Spring Harbor Perspect Med 2020; 10(3): a037093.2.Aleman S, Rahbin N, Weiland O, Davidsdottir L, Hedenstierna M, Rose N, et al. A risk for hepatocellular carcinoma persists long-term after sustained virologic response in patients with hepatitis C–associated liver cirrhosis. Clin Infect Dis 2013; 57(2): 230-6.
3.Patil S, Rao A, Pathak P, Kurle S, Mane A, Nirmalkar A, et al. Unsterile injection equipment associated with HIV outbreak and an extremely high prevalence of HCV—A case-control investigation from Unnao, India. PloS one 2020; 15(12): e0243534.
4.Sierpińska L. Circumstances of infection with HCV in selected Polish provinces. J Edu Health Sport 2021; 11(2): 41-52.
5.Alkareemy EAR, El-Din Hafiz MZ, Ahmed SAM, Abd el Aal Ahmed ASA. Effect of Treating Chronic Hepatitis C Infection with Direct-Acting Antivirals on The Risk of Recurrence Hepatocellular Carcinoma. Egypt J Hospital Med 2020; 79(1); 362-8.
6.De Luca A, Bianco C, Rossetti B. Treatment of HCV infection with the novel NS3/4A protease inhibitors. Curr Opin Pharmacol 2014; 18: 9-17.
7.Götte M, Feld JJ.Direct-acting antiviral agents for hepatitis C: structural and mechanistic insights. Nat Rev Gastroenterol Hepatol 2016; 13(6): 338-51.
8.J Feld J. Direct-acting antivirals for hepatitis C virus (HCV): the progress continues. Curr Drug Targ 2017; 18(7): 851-62.
9.Waheed Y, Siddiq M. Elimination of hepatitis from Pakistan by 2030: is it possible? Hepatoma Res 2018; 4(1): 45.
10.Khaliq S, Raza SM. Current status of direct acting antiviral agents against hepatitis C virus infection in Pakistan. Medicina 2018; 54(5):80.
11.Keating GM. Daclatasvir: a review in chronic hepatitis C. Drugs 2016; 76(14): 1381-91.
12.Ahmed MM, Abdel-Gawad M, Elkady A. Effect of Direct Acting Anti-Hepatitis C Drugs on the Heart. Egypt J Hospital Med 2021; 82(1): 106-14.
13.Shousha HI, Said M, ElAkelW, ElShafei A, Esmat G, Waked E, et al. Assessment of facility performance during mass treatment of chronic hepatitis C in Egypt: enablers and obstacles. J Infect Public Health 2020; 13(9): 1322-9.
14.Waziry R, Hajarizadeh B, Grebely J, Amin J, Law M, Danta M, et al. Hepatocellular carcinoma risk following direct-acting antiviral HCV therapy: a systematic review, meta-analyses, and meta-regression. J Hepatol 2017; 67(6): 1204-12.
15.van der Meer AJ, Maan R, Veldt BJ, Feld JJ, Wedemeyer H, Dufour JF, et al. Improvement of platelets after SVR among patients with chronic HCV infection and advanced hepatic fibrosis. J Gastroenterol Hepatol 2016; 31(6): 1168-76.
16.Jensen SB, Fahnøe U, Pham LV, Serre SBN, Tang Q, Ghanem L, et al. Evolutionary pathways to persistence of highly fit and resistant hepatitis C virus protease inhibitor escape variants. Hepatol 2019; 70(3): 771-87.
17.Buti M, Riveiro-Barciela M, Esteban R. Management of direct-acting antiviral agent failures. J Hepatol 2015; 63(6): 1511-22.
18.Pol S, Corouge M, Vallet-Pichard A. Daclatasvir–sofosbuvir combination therapy with or without ribavirin for hepatitis C virus infection: from the clinical trials to real life. Hepat Med 2016; 8(3): 21-6.
19.Gill M. Outcomes of daclatasvir+ sofosbuvir+ riba in HEP C G3 patients who relapsed with SOF+ RIBA combination therapy. J Hepatol 2017; 1(66): S725.
20.Petruzziello A, Marigliano S, Loquercio G, Coppola N, Piccirillo M, Leongito M, et al. Hepatitis C Virus (HCV) genotypes distribution among hepatocellular carcinoma patients in Southern Italy: a three year retrospective study. Infect Agent Cancer 2017; 12(1): 1-8.
21.Kutala BK, Mouri F, Castelnau C, Bouton V, Giuily N, Boyer N, et al. Efficacy and safety of sofosbuvir-based therapies in patients with advanced liver disease in a real-life cohort. Hepatic Med 2017; 9: 67.