Dr. Ahmad Shahzad
Founder | Lyallpur Diabetes Foundation
Consultant Diabetologist | Educator | Advocate for Preventive Care
Diabetes is a complicated metabolic condition that could cause severe health problems besides high blood sugar. Oxidative stress is one of the major contributors to these complications, as this is a condition of imbalance in the body between free radicals and antioxidants. It is also proven that oxidative stress contributes significantly to the destruction of blood vessels, nerves, kidneys and other organs and thus is directly connected to diabetic complications. Understanding the role of oxidative stress in diabetic complications is essential for developing better strategies to prevent, manage, and treat long-term effects of the disease.
What is Oxidative Stress?
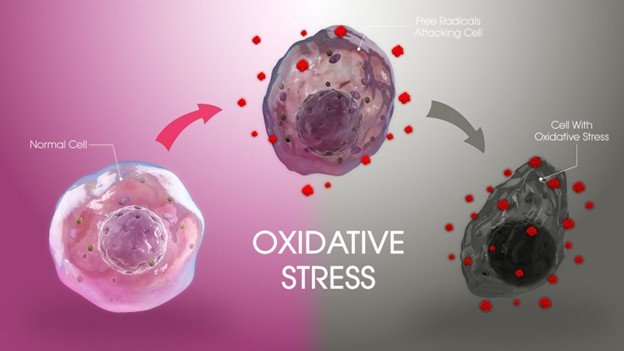
Oxidative stress refers to a situation in which the formation and accumulation of reactive oxygen species (ROS), or free radicals, and the capacity to eliminate the reactive species or the damage caused by them is imbalanced. The result of this imbalance is cell and tissue damage since the surplus free radicals can damage vital structures in cells like proteins, lipids, and DNA. Although ROS are necessary in normal cellular activities such as signaling, immune defense, high levels of ROS because of environmental factors or metabolic imbalance led to oxidative stress, which causes aging and many diseases, including cancer, diabetes, cardiovascular and neurodegeneration diseases.
This imbalance arises when the free radicals exceed the antioxidant capacity of antioxidants, the molecules that could stabilize free radicals by donating electrons. Pollution, UV radiation, smoking and some drugs are some of the factors that can initiate oxidative stress. Enzymatic antioxidants like superoxide dismutase, catalase and glutathione peroxidase are used by the body to defend itself, yet, when these systems become overburdened, cell damage occurs.
Oxidative Stress and Diabetes: The Connection
Diabetes is closely related to oxidative stress in a complex way where oxidative stress is essential in pathogenesis, progression, and complications of diabetes. Prolonged high blood glucose (hyperglycemia) and mitochondrial dysfunction in diabetes, especially in type 2 diabetes, cause excess production of reactive oxygen species (ROS). Such surges in ROS exacerbate oxidative stress, which harms pancreatic beta cells that produce insulin, disabling their activity and leading to cell death, decreasing insulin production. Several other causes also lead to insulin resistance, including oxidative stress, when the cells of the body cannot react to insulin, further disrupting the glucose regulation.
Besides, oxidative stress contributes significantly to diabetic complications involving various organs. It harms the vascular system by fostering inflammation and endothelial dysfunction resulting in microvascular complications such as diabetic retinopathy (eye destruction), nephropathy (kidney destruction) and neuropathy (nerve destruction). It also leads to macrovascular problems, which are a risk factor of developing cardiovascular diseases in diabetics. The permanent oxidative injury may perpetuate a deleterious hyperglycemic memory, continuing tissue dysfunction despite normalization of blood glucose.
Antioxidant therapies, lifestyle activities, including diet and exercise, as well as careful blood sugar control, are strategies to counter oxidative stress in diabetes. Nevertheless, the mechanisms remain unclear, and more studies are required to maximize the therapies that address oxidative stress to prevent and manage diabetic complications.
Mechanisms Behind Oxidative Stress in Diabetes
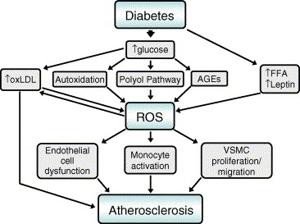
Oxidative stress mechanisms in diabetes entail a series of interacting events that cause an overproduction of reactive oxygen species (ROS) and dysfunction of antioxidant responses and play a direct role in the pathogenesis and complications of diabetes.
Sources of ROS in Diabetes:
Mitochondria during cellular respiration are the major source of oxidative stress in diabetes. Hyperglycemia (high blood sugar) leads to excess glucose metabolism via glycolysis and Krebs cycle leading to high levels of electron donors (NADH and FADH2) overloading the electron transport chain. This leads to loss of electrons to oxygen to form superoxide radicals (O2- -) and other ROS like hydrogen peroxide (H2O2) and hydroxyl radicals (- OH). Moreover, there is hyperactivity of enzymatic systems such as NADPH oxidase, which increases the production of ROS in endothelial cells, smooth muscle cells, and fibroblasts especially when hyperglycemia occurs.
Impaired Antioxidant Defense:
Diabetes is known to break the equilibrium by reducing the activity of protective antioxidant enzymes such as superoxide dismutase (SOD), catalase and glutathione peroxidase (GSH-Px). Decreased antioxidants result in accumulating ROS with destruction of cells.
Molecular Pathways Amplifying Oxidative Stress:
Protein Glycation and lipid peroxidation: Non-enzymatic glycation of proteins and lipid peroxidation leads to the production of free radicals and advanced glycation end products (AGEs), which further enhance the production of ROS and inflammation.
- Polyol Pathway Activation: Aldose reductase converts excess glucose into sorbitol and depletes NADPH and weakens the ability to regenerate antioxidants.
- Protein Kinase C (PKC) Activation: Hyperglycemia triggers PKC, which promotes NADPH oxidase, elevates inflammatory cytokines and modulates gene expression of cell damage and vascular dysfunction.
- Hexosamine Pathway: Enhanced activity of this pathway alters transcription factors by O-linked glycosylation and impacts diabetic complication related genes.
- Chronic Inflammation: ROS stimulate inflammatory transcription factors (e.g., NF-kB) which leads to driving cytokine production and maintaining a vicious cycle of ROS production.
Impact on Insulin and Beta Cells:
The low antioxidant capacity of ROS leads to insulin secretion and beta cell apoptosis as these molecules cause oxidative damage to pancreatic beta cells. Furthermore, oxidative stress causes insulin resistance by disrupting the insulin signaling that occurs in insulin metabolic dysfunction that is typical of type 2 diabetes.
Contribution to Diabetic Complications
Oxidative stress injures endothelial cells, resulting in vascular dysfunction, microvascular (retinopathy, nephropathy, neuropathy) and macrovascular (cardiovascular disease) complications.
Oxidative Stress in Diabetic Complications
Oxidative stress has been at the center of the development and progress of diabetic complications, micro and macro vascular. Chronic elevated blood sugar in diabetes leads to mitochondrial overproduction of reactive oxygen species (ROS) in endothelial cells and other tissues, resulting in several harmful effects that cause diabetic complications.
Key pathways activated by oxidative stress in diabetes complications include:
- Polyol Pathway Flux: Surplus glucose is metabolized to sorbitol by aldose reductase, depleting NADPH, decreasing antioxidant regeneration, and predisposing cells to oxidative damage.
- Advanced Glycation End-Products (AGEs): High glucose enhances the development of AGEs which bind to their receptors (RAGE), leading to the production of ROS, inflammation and tissue damage observed in diabetic nephropathy, retinopathy and neuropathy.
- Activation of Protein Kinase C (PKC): ACTH hyperglycemia-activated PKC isoforms upregulate ROS, inflammation, vascular permeability, and abnormal blood flow, which add to vascular complications.
- Hexosamine Pathway: Overflow of glucose flux causes changes in transcription factors that control fibrosis and inflammatory genes, exacerbating tissue injury.
- Direct Enzyme Inactivation: ROS directly inactivates protective against atherosclerosis, including endothelial nitric oxide synthase (eNOS) and prostacyclin synthase, disrupting vascular activity.
Activation of transcription factors such as NF-KB through oxidative stress results in a vicious cycle of cytokine release and immune cell recruitment that exacerbates vascular and tissue injury.
Most heavily impacted organs are kidneys (diabetic nephropathy), eyes (retinopathy), nerves (neuropathy), and the cardiovascular system, which cause greater risk of kidney failure, blindness, neuropathic pain, and heart disease. Epigenetic changes which persist pro-inflammatory gene activity in the face of controlled blood sugar, or so-called metabolic memory, are also caused by oxidative stress.
Superoxide dismutase, catalase, and glutathione peroxidase are the antioxidant defenses that are also impaired in diabetes, thus worsening oxidative damage.
You may also like to read: Importance of Foot Care in Preventing Diabetic Foot Ulcers
Role in Beta-Cell Dysfunction and Insulin Resistance
There are multiple ways that oxidative stress contributes to beta-cell dysfunction and insulin resistance in diabetes:
Beta-Cell Dysfunction
- Susceptibility: Pancreatic beta cells possess low intrinsic antioxidant capacity and are therefore extremely susceptible to oxidative stresses produced by overproduction of reactive oxygen species (ROS) under hyperglycemic and inflammatory conditions.
- Damage and Death: Overproduction of ROS results in mitochondrial dysfunction and endoplasmic reticulum (ER) stress in beta cells, leading to cell apoptosis and insulin secretion.
- Loss of Identity: Oxidative stress results in loss of important beta-cell maturity transcription factors like MAFA and PDX1 to cause beta-cell dedifferentiation and decreased insulin synthesis.
- Inflammation: ROS activate immune systems through pro-inflammatory cytokines (e.g., IFN-7, IL-1 1, TNF-7) that contribute to additional beta cell death, particularly in diabetes type 1.
- Antioxidant Downregulation: Downregulation of endogenous antioxidants such as superoxide dismutase (SOD2) enhances oxidative beta cell injury.
- Protective Pathways: FOXO1 to the nucleus is a defense to oxidative stress that can be overwhelmed in chronic conditions.
Insulin Resistance
· Disruption of the Insulin Signaling: ROS trigger serine/threonine kinases including JNK, p38 MAPK, and IKK that phosphorylate insulin receptor substrates (IRS) on serine residues and disrupt their function in relaying insulin signals in cells like muscle and adipose tissue.
· PI3K Disruption: Oxidative stress interferes with translocation and activation of PI3K, an insulin signaler, facilitating insulin resistance.
· PI3K Disruption: Oxidative stress interferes with translocation and activation of PI3K, an insulin signaler, facilitating insulin resistance.
· Inflammatory Pathways: ROS stimulate inflammatory pathways (NF-KB), prolonging the systemic inflammation thereby worsening further insulin insensitivity.
· Redox Imbalance: The imbalance between the production of ROS and antioxidant defense is one of the causes of disturbed metabolism and desensitization of insulin receptors in peripheral tissues.
Strategies to Reduce Oxidative Stress in Diabetes
Oxidative stress reduction strategies in diabetes involve a combination of lifestyle change, antioxidant therapy, and appropriate management of the disease to regain redox balance and alleviate cellular injury:
Lifestyle Modifications
- Diet: The Mediterranean diet is rich in natural antioxidants in fruits, vegetables, whole grains, olive oil, nuts, and fish, and has been shown to be effective in improving antioxidant enzyme activity and reducing oxidative stress markers. The activation of NRF2 pathway by key phytochemicals upregulates the protective antioxidant genes such as glutathione peroxidase and superoxide dismutase. Low-calorie diets decrease the variability of blood glucose and the production of reactive oxygen species (ROS).
- Exercise: Mediate aerobic exercise (e.g., walking, cycling) enhances mitochondrial activity and increases endogenous antioxidant enzymes, alleviating oxidative injury in diabetic individuals. Resistance training and high-intensity interval training (HIIT) also have potential in improving antioxidant defenses and glycemic control, but individual health status and age can vary in response. Moderate workouts will avoid the overload of ROS that may exacerbate stress.
- Stress Management and Sleep: Mindfulness, yoga, and sufficient sleep, lower systemic inflammation and cortisol which otherwise contribute to increased oxidative stress and insulin resistance.
Antioxidant Therapies
- Supplementation: Alpha-lipoic acid, coenzyme Q10, glutathione and vitamins C and E have also been studied to restore insulin sensitivity and prevent oxidative damage, although the clinical benefit is inconsistent.
- Targeted Therapeutics: New therapeutic strategies target mitochondria-targeted antioxidants and activators of transcription factors such as NRF2 that control antioxidant defenses. It is also being investigated to inhibit NADPH oxidase (NOX) enzymes, which are key sources of ROS.
- Nanotechnology and Microbiome: new directions involve nanocarrier-delivered antioxidants to pancreatic islands or atherosclerotic foci, or the regulation of the gut microbiota to ease overall oxidative stress in the body.
Effective Diabetes Management
- Blood Sugar Control: Tight glycemic control by diet, medication and monitoring mitigate excessive ROS generation in response to hyperglycemia and oxidative stress.
- Medications: some antidiabetic drugs such as metformin and GLP-1 receptor agonists are antioxidants that minimize oxidative stress beyond the effect of glucose.
Final Thoughts
Summing up, oxidative stress plays a key role in most diabetic complications, including nerve and kidney damage and cardiovascular disease. It impairs the long-term outcomes of diabetes by destabilizing the cellular balance and triggering inflammation. Addressing the role of oxidative stress in diabetic complications through lifestyle changes, antioxidant support, and targeted therapies offers a promising path to protect patients’ health and improve quality of life.